|
In other books or articles, you may sometimes see benzene or a phenyl group drawn with a circle inside the hexagon, either solid or dashed, as a way of drawing a resonance hybrid. This is the convention that will be used for the most part in this book. Usually, derivatives of benzene (and phenyl groups, when the benzene ring is incorporated into a larger organic structure) are depicted with only one resonance contributor, and it is assumed that the reader understands that resonance hybridization is implied. 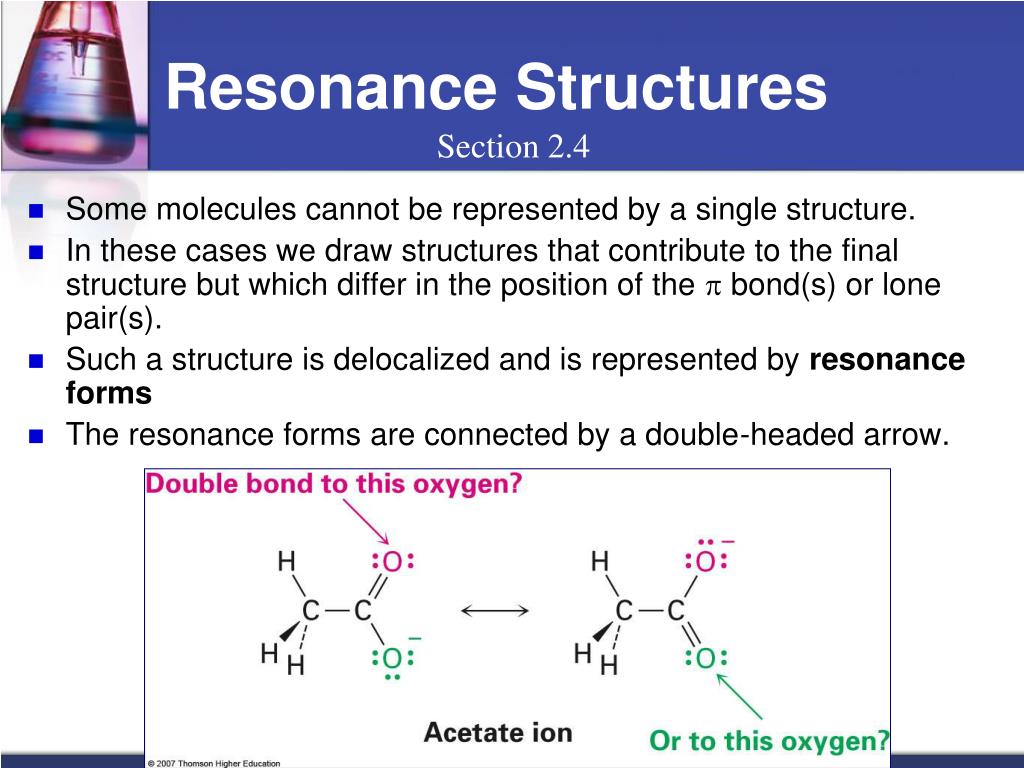
Furthermore, the double-headed resonance arrow does NOT mean that a chemical reaction has taken place. Caution! It is very important to be clear that in drawing two (or more) resonance contributors, we are not drawing two different molecules: they are simply different depictions of the exact same molecule. Rather, at all moments, the molecule is a combination, or resonance hybrid of both A and B. The depiction of benzene using the two resonance contributors A and B in the figure above does not imply that the molecule at one moment looks like structure A, then at the next moment shifts to look like structure B. Nevertheless, use of the curved arrow notation is an essential skill that you will need to develop in drawing resonance contributors. In the drawing of resonance contributors, however, this electron ‘movement’ occurs only in our minds, as we try to visualize delocalized pi bonds. A few chapters from now when we begin to study organic reactions - a process in which electron density shifts and covalent bonds between atoms break and form - this ‘curved arrow notation’ will become extremely important in depicting electron movement. Each of these arrows depicts the ‘movement’ of two pi electrons. In order to make it easier to visualize the difference between two resonance contributors, small, curved arrows are often used. By convention, resonance contributors are linked by a double-headed arrow, and are sometimes enclosed by brackets: These two drawings are an example of what is referred to in organic chemistry as resonance contributors: two or more different Lewis structures depicting the same molecule or ion that, when considered together, do a better job of approximating delocalized pi-bonding than any single structure. This is because they imply, together, that the carbon-carbon bonds are not double bonds, not single bonds, but about halfway in between. The two alternative drawings, however, when considered together, give a much more accurate picture than either one on its own. Both ways of drawing the molecule are equally acceptable approximations of the bonding picture for the molecule, but neither one, by itself, is an accurate picture of the delocalized pi bonds. Which way is correct? There are two simple answers to this question: 'both' and 'neither one'. 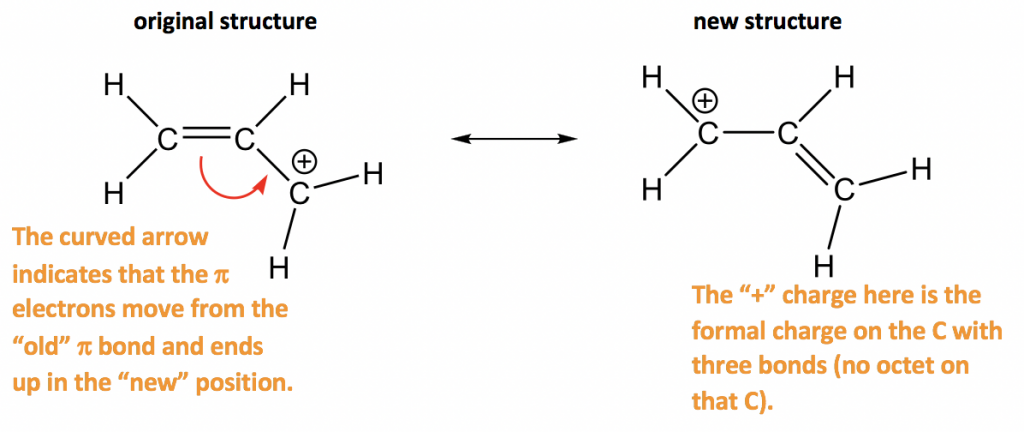
If we were to draw the structure of an aromatic molecule such as 1,2-dimethylbenzene, there are two ways that we could draw the double bonds:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
